Nano-Science Center Seminar: Birger Lindberg Møller
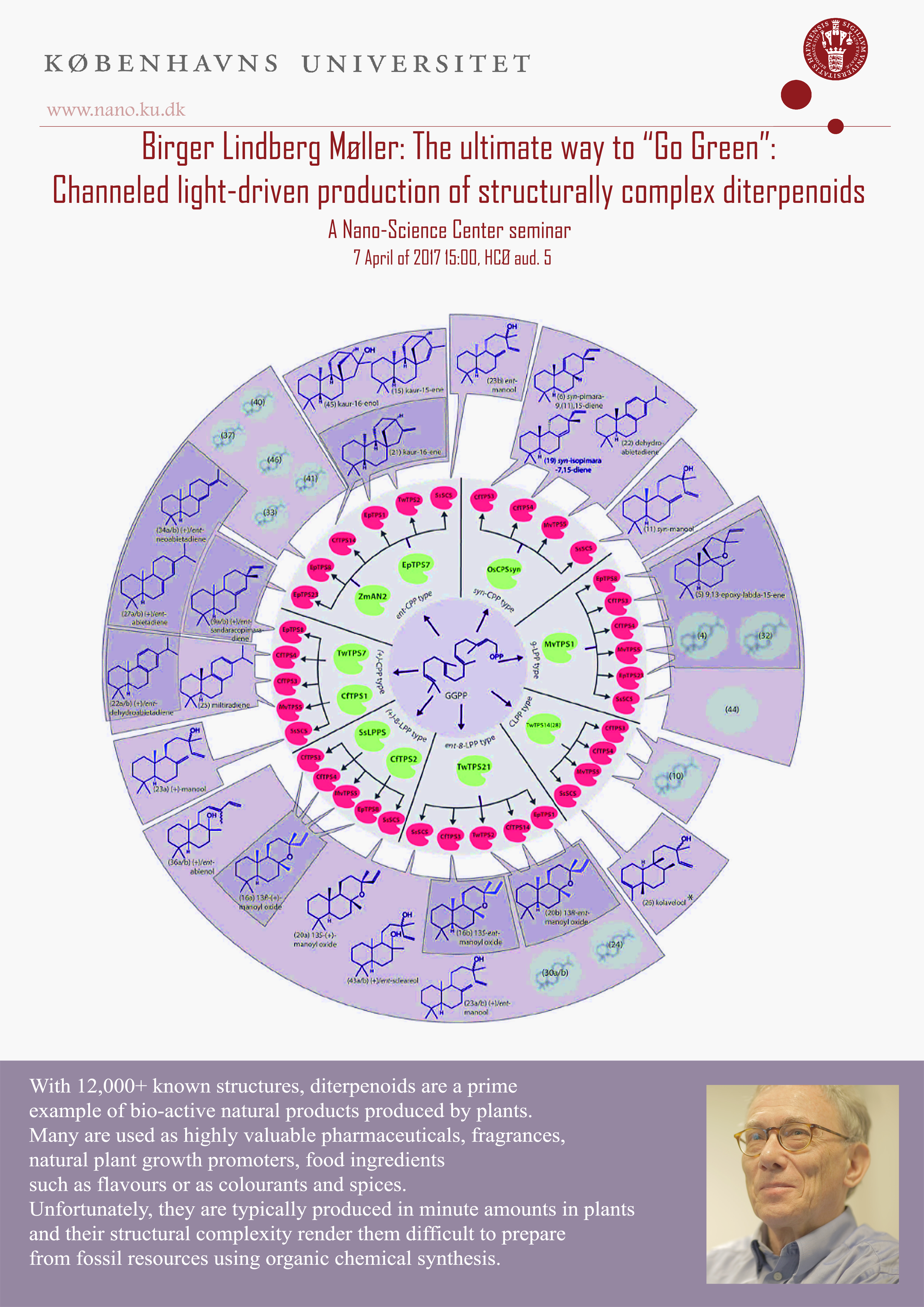
Terpenoid synthases, cytochrome P450s and acyl transferases are key multienzyme families involved in diterpenoid synthesis. Using mass spec-based imaging of the target plant tissue, tracer studies, single cell-type based metabolomics and transcriptomics, functional characterization of gene candidates using transient expression in tobacco and LC-MS-NMR-based structural identification, elucidation of even highly complex biosynthetic pathways is now possible within a reasonably short time frame. Terpenoid metabolism is modular right from assembly of the C5 building blocks to the final, structurally complex diterpenoid. Using the approaches of synthetic biology for combinatorial biosynthesis, the functional modules may be assembled in new combinations to expand the landscape of diterpenoid structural diversity into new-to-nature structures. Our recent elucidation of the entire pathway for forskolin will be presented. Forskolin is a cyclic AMP booster approved for treatment of glaucoma but also used as a weight loss aid. The forskolin pathway is being used as test model system for large-scale, light-driven production of high-value diterpenoids following targeting of the pathway to the photosynthetic thylaloid membrane using cyanobacteria or moss as photosynthetic production hosts grown in contained photo-bioreactors.
