Electrochemical CO2 Reduction: A Classification Problem
Alexander Bagger, Wen Ju, Ana Sofia Varela, Peter Strasser and Jan Rossmeisl
Manuscript accepted in ChemPhysChem 10.1002/cphc.201700736DOI: http://dx.doi.org/10.1002/cphc.201700736Description
In this work we propose four non-coupled binding energies of intermediates as descriptors, or 'genes', for predicting the product distribution in CO2 electroreduction. Simple reactions can be understood by the Sabatier principle (catalytic activity vs. one descriptor), while more complex reactions tend to give multiple very different products and consequently the product selectivity is a more complex property to understand. We approach this, as a logistical classification problem, by grouping metals according to their major experimental product from CO2 electroreduction: H2, CO, formic acid and beyond CO* (hydrocarbons or alcohols). We compare the groups in terms of multiple binding energies of intermediates calculated by density functional theory. Here we find three descriptors to explain the grouping: the adsorption energies of H*, COOH* and CO*. To further classify products beyond CO*, we carry out formaldehyde experiments on Cu, Ag and Au and combine these results with the literature to group and differentiate alcohol or hydrocarbon products. We find that the oxygen binding (adsorption energy of CH3O*) is an additional descriptor to explain the alcohol formation in reduction processes. Finally, the adsorption energy of the four intermediates, H*, COOH*, CO* and CH3O*, can be used to differentiate, group and explain products in electrochemical reduction processes involving CO2, CO and carbon-oxygen compounds.
The Database
Download metal databaseDownload molecule databaseKey-value pairs: Description
Given and used in python scripts, but also listed below here.
Key words for metals large database:
Metal: 'Cu','Ag','Au','Ir','Pt','Pd','Rh','Sn','In','Pb','Cd','Tl','Ru','Ni', 'Fe', 'Ti','Hg','Zn'
ads: 'COOH', 'H','CO', 'Clean', 'HCOO', 'CH3O', 'CH2OH''
Key words for molecule database:
Molecule: 'CO2', 'CO', 'H2', 'CH2O'
A combined plot of the three proposed descriptors in CO2 electroreduction towards H2, CO and HCOOH (formic acid) for metals.
Download script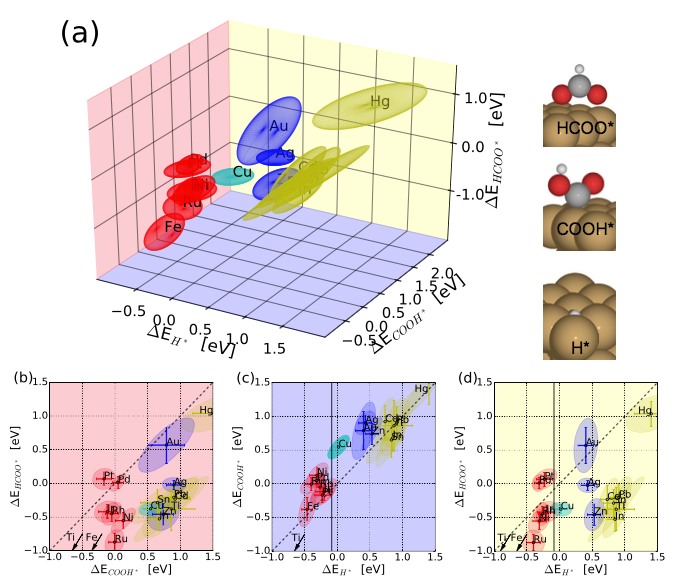 (a) the three dimensional space of descriptors are plotted with the coupled binding energy ellipsoids that represent the first σ confidence interval of the calculations with respect to Au, while the Au error ellipsoid is given with respect to the slab and the gas phase (see script how this is carried out). Each background plane has been giving a color to match the 2d projections of energies and ellipses in (b), (c) and (d) for the ∆E HCOO* vs ∆E COOH* , ∆E COOH* vs. ∆E H* and ∆E HCOO* vs. ∆E H* planes, respectively. In (b), (c) and (d) the solid black lines shown the thermodynamic line (see script for value) and the diagonal dashed black lines indicate the relative stability of the two adsorbates.
(a) the three dimensional space of descriptors are plotted with the coupled binding energy ellipsoids that represent the first σ confidence interval of the calculations with respect to Au, while the Au error ellipsoid is given with respect to the slab and the gas phase (see script how this is carried out). Each background plane has been giving a color to match the 2d projections of energies and ellipses in (b), (c) and (d) for the ∆E HCOO* vs ∆E COOH* , ∆E COOH* vs. ∆E H* and ∆E HCOO* vs. ∆E H* planes, respectively. In (b), (c) and (d) the solid black lines shown the thermodynamic line (see script for value) and the diagonal dashed black lines indicate the relative stability of the two adsorbates.
